Chapter 8. Membrane Transport

Chapter Outline
- 8.1 Membrane Components and Structure
- 8.2 Passive Transport
- 8.3 Active Transport
- 8.4 Bulk Transport
Introduction
The plasma membrane, which is also called the cell membrane, has many functions, but the most basic one is to define the borders of the cell and keep the cell functional. The plasma membrane is selectively permeable. This means that the membrane allows some materials to freely enter or leave the cell, while other materials cannot move freely, but require the use of a specialized structure, and occasionally, even energy investment for crossing.
8.1 | Membrane Components and Structure
Learning Objectives
By the end of this section, you will be able to:
- Understand the fluid mosaic model of cell membranes.
- Describe the functions of phospholipids, proteins, and carbohydrates in membranes.
- Discuss membrane fluidity.
A cell’s plasma membrane defines the cell, outlines its borders, and determines the nature of its interaction with its environment (see Table 8.1 for a summary). Cells exclude some substances, take in others, and excrete still others, all in controlled quantities. The plasma membrane must be very flexible to allow certain cells, such as red blood cells and white blood cells, to change shape as they pass through narrow capillaries. These are the more obvious functions of a plasma membrane. In addition, the surface of the plasma membrane carries markers that allow cells to recognize one another, which is vital for tissue and organ formation during early development, and which later plays a role in the “self” versus “non-self” distinction of the immune response.
Among the most sophisticated functions of the plasma membrane is the ability to transmit signals by means of complex, integral proteins known as receptors. These proteins act both as receivers of extracellular inputs and as activators of intracellular processes. These membrane receptors provide extracellular attachment sites for effectors like hormones and growth factors, and they activate intracellular response cascades when their effectors are bound. Occasionally, receptors are hijacked by viruses (HIV, human immunodeficiency virus, is one example) that use them to gain entry into cells, and at times, the genes encoding receptors become mutated, causing the process of signal transduction to malfunction with disastrous consequences.
8.1.1 Fluid Mosaic Model
The existence of the plasma membrane was identified in the 1890s, and its chemical components were identified in 1915. The principal components identified at that time were lipids and proteins. The first widely accepted model of the plasma membrane’s structure was proposed in 1935 by Hugh Davson and James Danielli; it was based on the “railroad track” appearance of the plasma membrane in early electron micrographs. They theorized that the structure of the plasma membrane resembles a sandwich, with protein being analogous to the bread, and lipids being analogous to the filling. In the 1950s, advances in microscopy, notably transmission electron microscopy (TEM), allowed researchers to see that the core of the plasma membrane consisted of a double, rather than a single, layer. A new model that better explains both the microscopic observations and the function of that plasma membrane was proposed by S.J. Singer and Garth L. Nicolson in 1972.
The explanation proposed by Singer and Nicolson is called the fluid mosaic model. The model has evolved somewhat over time, but it still best accounts for the structure and functions of the plasma membrane as we now understand them. The fluid mosaic model describes the structure of the plasma membrane as a mosaic of components—including phospholipids, cholesterol, proteins, and carbohydrates—that gives the membrane a fluid character. Plasma membranes range from 5 to 10 nm in thickness. For comparison, human red blood cells, visible via light microscopy, are approximately 8 µm wide, or approximately 1,000 times wider than a plasma membrane. The membrane does look a bit like a sandwich (Figure 8.2).
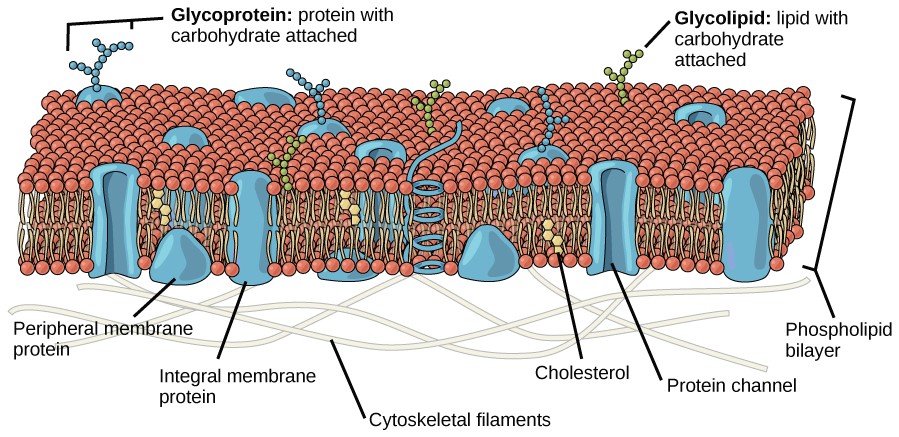
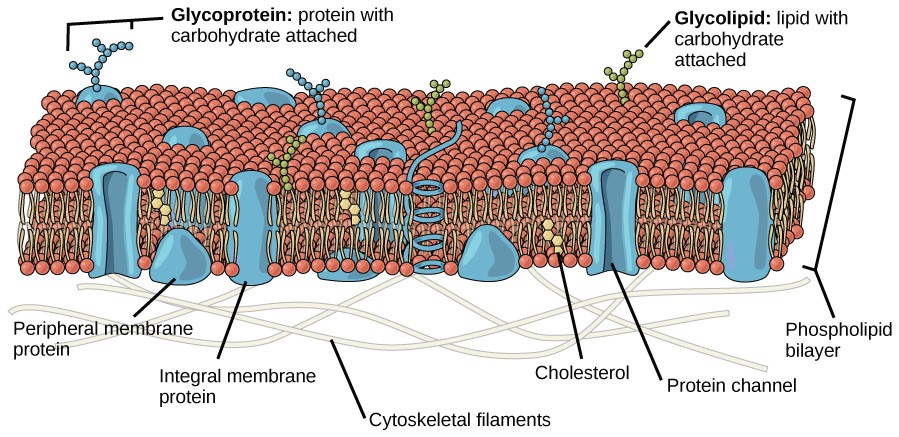
The principal components of a plasma membrane are lipids, proteins, and carbohydrates. The lipids include phospholipids and cholesterol Proteins either float in the bilayer or are attached to one side or the other of it. Carbohydrate chains are attached to the proteins and lipids on the outside surface of the membrane. The proportions of proteins, lipids, and carbohydrates in the plasma membrane vary with cell type, but for a typical human cell, protein accounts for about 50 percent of the composition by mass, lipids account for about 40 percent of the composition by mass, with the remaining 10 percent of the composition by mass being carbohydrates.
Phospholipids
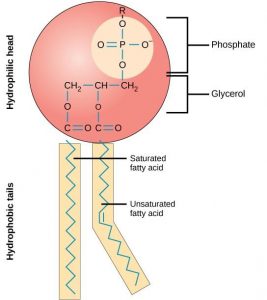
The main fabric of the membrane is composed of amphiphilic phospholipid molecules. Recall from chapter 4 that a phospholipid is a molecule consisting of glycerol, two fatty acids, and a phosphate-linked head group (Figure 8.3) . The hydrophilic “head” of these molecules are in contact with the aqueous fluid both inside and outside the cell. The hydrophobic “tails” face each other in the inside of the bilayer. Therefore, phospholipids form an excellent two-layer cell membrane that separates fluid within the cell from the fluid outside of the cell (Figure 8.2).
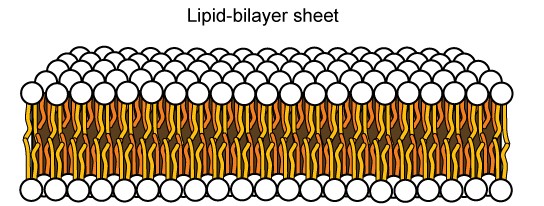
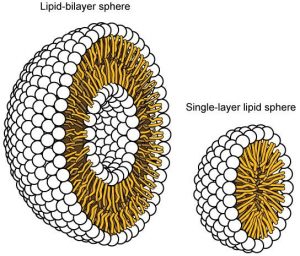
The amphipathic nature of phospholipids is vital to the structure of a plasma membrane because, in water, phospholipids automatically become arranged with their hydrophobic tails facing each other and their hydrophilic heads facing out. In this way, they form a lipid bilayer—a barrier composed of a double layer of phospholipids that separates the water and other materials on one side of the barrier from the water and other materials on the other side (Figure 8.4 top). In fact, phospholipids heated in an aqueous solution tend to spontaneously form small spheres or droplets called micelles, with their hydrophilic heads forming the exterior and their hydrophobic tails on the inside (Figure 8.4 bottom).
Proteins
Proteins make up the second major component of plasma membranes. Integral proteins are, as their name suggests, integrated completely into the membrane structure, and their hydrophobic membrane-spanning regions interact with the hydrophobic region of the the phospholipid bilayer (Figure 8.2). Single-pass integral membrane proteins usually have a hydrophobic transmembrane segment that consists of 20–25 amino acids. Some span only part of the membrane— associating with a single layer—while others stretch from one side of the membrane to the other, and are exposed on either side. Since they cross the membrane, these are often called transmembrane proteins.
Some complex integral proteins are composed of up to 12 segments, which are extensively folded and embedded in the membrane (Figure 8.5). This type of protein has a hydrophilic region or regions, and several hydrophobic regions. This arrangement of regions of the protein tends to orient the protein alongside the phospholipids, with the hydrophobic region of the protein adjacent to the tails of the phospholipids and the hydrophilic region or regions of the protein protruding from the membrane and in contact with the cytosol or extracellular fluid.
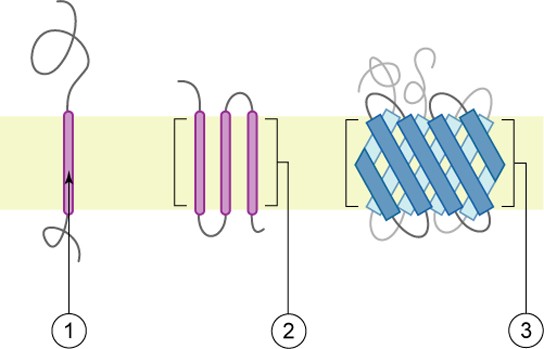
Peripheral proteins are found on the exterior and interior surfaces of membranes, attached either to integral proteins or to phospholipids. Peripheral proteins, along with integral proteins, may serve as enzymes, as structural attachments for the fibers of the cytoskeleton, or as part of the cell’s recognition sites. These are sometimes referred to as “cell-specific” proteins. The body recognizes its own proteins and attacks foreign proteins associated with invasive pathogens.
Carbohydrates
Carbohydrates are the third major component of plasma membranes. They are always found on the exterior surface of cells and are bound either to proteins (forming glycoproteins) or to lipids (forming glycolipids) (Figure 8.2). These carbohydrate chains may consist of 2–60 monosaccharide units and can be either straight or branched. Along with peripheral proteins, carbohydrates form specialized sites on the cell surface that allow cells to recognize each other. These sites have unique patterns that allow the cell to be recognized, much the way that the facial features unique to each person allow him or her to be recognized. This recognition function is very important to cells, as it allows the immune system to differentiate between body cells (called “self”) and foreign cells or tissues (called “non-self”). Similar types of glycoproteins and glycolipids are found on the surfaces of viruses and may change frequently, preventing immune cells from recognizing and attacking them.
These carbohydrates on the exterior surface of the cell—the carbohydrate components of both glycoproteins and glycolipids—are collectively referred to as the glycocalyx (meaning “sugar coating”). The glycocalyx is highly hydrophilic and attracts large amounts of water to the surface of the cell. This aids in the interaction of the cell with its watery environment and in the cell’s ability to obtain substances dissolved in the water. As discussed above, the glycocalyx is also important for cell identification, self/non-self determination, and embryonic development, and is used in cell-cell attachments to form tissues.
![]()
How Viruses Infect Specific Organs
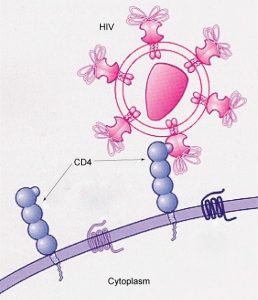
Glycoprotein and glycolipid patterns on the surfaces of cells give many viruses an opportunity for infection. HIV and hepatitis viruses infect only specific organs or cells in the human body. HIV is able to penetrate the plasma membranes of a subtype of lymphocytes called T-helper cells, as well as some monocytes and central nervous system cells. The hepatitis virus attacks liver cells.
These viruses are able to invade these cells, because the cells have binding sites on their surfaces that are specific to and compatible with certain viruses (Figure 8.6). Other recognition sites on the virus’s surface interact with the human immune system, prompting the body to produce antibodies. Antibodies are made in response to the antigens or proteins associated with invasive pathogens, or in response to foreign cells, such as might occur with an organ transplant. These same sites serve as places for antibodies to attach and either destroy or inhibit the activity of the virus. Unfortunately, these recognition sites on HIV change at a rapid rate because of mutations, making the production of an effective vaccine against the virus very difficult, as the virus evolves and adapts. A person infected with HIV will quickly develop different populations, or variants, of the virus that are distinguished by differences in these recognition sites. This rapid change of surface markers decreases the effectiveness of the person’s immune system in attacking the virus, because the antibodies will not recognize the new variations of the surface patterns. In the case of HIV, the problem is compounded by the fact that the virus specifically infects and destroys cells involved in the immune response, further incapacitating the host.
8.1.2 Membrane Fluidity
The mosaic characteristic of the membrane, described in the fluid mosaic model, helps to illustrate its nature. The integral proteins and lipids exist in the membrane as separate but loosely attached molecules. These resemble the separate, multicolored tiles of a mosaic picture, and they float, moving somewhat with respect to one another. The membrane is not like a balloon, however, that can expand and contract; rather, it is fairly rigid and can burst if penetrated or if a cell takes in too much water. However, because of its mosaic nature, a very fine needle can easily penetrate a plasma membrane without causing it to burst, and the membrane will flow and self-seal when the needle is extracted.
The mosaic characteristics of the membrane explain some but not all of its fluidity. There are two other factors that help maintain this fluid characteristic. One factor is the nature of the phospholipids themselves. In their saturated form, the fatty acids in phospholipid tails are saturated with bound hydrogen atoms. There are no double bonds between adjacent carbon atoms. This results in tails that are relatively straight. In contrast, unsaturated fatty acids do not contain a maximal number of hydrogen atoms, but they do contain some double bonds between adjacent carbon atoms; a double bond results in a bend in the string of carbons of approximately 30 degrees (Figure 8.3).
Thus, if saturated fatty acids, with their straight tails, are compressed by decreasing temperatures, they press in on each other, making a dense and fairly rigid membrane. If unsaturated fatty acids are compressed, the “kinks” in their tails elbow adjacent phospholipid molecules away, maintaining some space between the phospholipid molecules. This “elbow room” helps to maintain fluidity in the membrane at temperatures at which membranes with saturated fatty acid tails in their phospholipids would “freeze” or solidify. The relative fluidity of the membrane is particularly important in a cold environment. A cold environment tends to compress membranes composed largely of saturated fatty acids, making them less fluid and more susceptible to rupturing. Many organisms (fish are one example) are capable of adapting to cold environments by changing the proportion of unsaturated fatty acids in their membranes in response to the lowering of the temperature.
Animals have an additional membrane constituent that assists in maintaining fluidity. Cholesterol, which lies alongside the phospholipids in the membrane, tends to dampen the effects of temperature on the membrane. Thus, this lipid functions as a buffer, preventing lower temperatures from inhibiting fluidity and preventing increased temperatures from increasing fluidity too much. Thus, cholesterol extends, in both directions, the range of temperature in which the membrane is appropriately fluid and consequently functional. Cholesterol also serves other functions, such as organizing clusters of transmembrane proteins into lipid rafts.
Table 8.1 The components and functions of the plasma membrane.
|
Component |
Location |
|
Phospholipid |
Main fabric of the membrane |
|
Cholesterol |
Attached between phospholipids and between the two phospholipid layers |
|
Integral proteins (for example, integrins) |
Embedded within the phospholipid layer(s). May or may not penetrate through both layers |
|
Peripheral proteins |
On the inner or outer surface of the phospholipid bilayer; not embedded within the phospholipids |
|
Carbohydrates (components of glycoproteins and glycolipids) |
Generally attached to proteins on the outside membrane layer |
![]()
Immunologist
The variations in peripheral proteins and carbohydrates that affect a cell’s recognition sites are of prime interest in immunology. These changes are taken into consideration in vaccine development. Many infectious diseases, such as smallpox, polio, diphtheria, and tetanus, were conquered by the use of vaccines.
dImmunologists are the physicians and scientists who research and develop vaccines, as well as treat and study allergies or other immune problems. Some immunologists study and treat autoimmune problems (diseases in which a person’s immune system attacks his or her own cells or tissues, such as lupus) and immunodeficiencies, whether acquired (such as acquired immunodeficiency syndrome, or AIDS) or hereditary (such as severe combined immunodeficiency, or SCID). Immunologists are called in to help treat organ transplantation patients, who must have their immune systems suppressed so that their bodies will not reject a transplanted organ. Some immunologists work to understand natural immunity and the effects of a person’s environment on it. Others work on questions about how the immune system affects diseases such as cancer. In the past, the importance of having a healthy immune system in preventing cancer was not at all understood.
To work as an immunologist, a PhD or MD is required. In addition, immunologists undertake at least 2–3 years of training in an accredited program and must pass an examination given by the American Board of Allergy and Immunology. Immunologists must possess knowledge of the functions of the human body as they relate to issues beyond immunization, and knowledge of pharmacology and medical technology, such as medications, therapies, test materials, and surgical procedures.
8.2 | Passive Transport
Learning Objectives
By the end of this section, you will be able to:
- Explain why and how passive transport occurs.
- Understand the processes of osmosis and diffusion.
- Define tonicity and describe its relevance to passive transport.
Plasma membranes must allow certain substances to enter and leave a cell, and prevent some harmful materials from entering and some essential materials from leaving. In other words, plasma membranes are selectively permeable—they allow some substances to pass through, but not others. If they were to lose this selectivity, the cell would no longer be able to sustain itself, and it would be destroyed. Some cells require larger amounts of specific substances than do other cells; they must have a way of obtaining these materials from extracellular fluids. This may happen passively, as certain materials move back and forth, or the cell may have special mechanisms that facilitate transport. Some materials are so important to a cell that it spends some of its energy, hydrolyzing adenosine triphosphate (ATP), to obtain these materials. All cells spend the majority of their energy to maintain an imbalance of sodium and potassium ions between the interior and exterior of the cell.
The most direct forms of membrane transport are passive. Passive transport is a naturally occurring phenomenon and does not require the cell to exert any of its energy to accomplish the movement. In passive transport, substances move from an area of higher concentration to an area of lower concentration. A physical space in which there is a range of concentrations of a single substance is said to have a concentration gradient.
8.2.1 Selective Permeability
Plasma membranes are asymmetric: the interior of the membrane is not identical to the exterior of the membrane. In fact, there is a considerable difference between the array of phospholipids and proteins between the two leaflets that form a membrane. On the interior of the membrane, some proteins serve to anchor the membrane to fibers of the cytoskeleton. There are peripheral proteins on the exterior of the membrane that bind elements of the extracellular matrix. Carbohydrates,
attached to lipids or proteins, are also found on the exterior surface of the plasma membrane. These carbohydrate complexes help the cell bind substances that the cell needs in the extracellular fluid. This adds considerably to the selective nature of plasma membranes (Figure 8.7).
Recall that plasma membranes are amphipathic: They have hydrophilic and hydrophobic regions. This characteristic helps the movement of some materials through the membrane and hinders the movement of others. Lipid-soluble material with a low molecular weight can easily slip through the hydrophobic lipid core of the membrane. Substances such as the fat- soluble vitamins A, D, E, and K readily pass through the plasma membranes in the digestive tract and other tissues. Fat-soluble drugs and hormones also gain easy entry into cells and are readily transported into the body’s tissues and organs. Molecules of oxygen and carbon dioxide have no charge and so pass through membranes by simple diffusion.
Polar substances present problems for the membrane. While some polar molecules connect easily with the outside of a cell, they cannot readily pass through the lipid core of the plasma membrane. Additionally, while small ions could easily slip through the spaces in the mosaic of the membrane, their charge prevents them from doing so. Ions such as sodium, potassium, calcium, and chloride must have special means of penetrating plasma membranes. Larger polar molecules, such as simple sugars and amino acids also need help with transport across plasma membranes.
8.2.2 Diffusion
Diffusion is a passive process of transport. A single substance tends to move from an area of high concentration to an area of low concentration until the concentration is equal across a space. You are familiar with diffusion of substances through the air. For example, think about someone opening a bottle of ammonia in a room filled with people. The ammonia gas is at its highest concentration in the bottle; its lowest concentration is at the edges of the room. The ammonia vapor will diffuse, or spread away, from the bottle, and gradually, more and more people will smell the ammonia as it spreads. Materials move within the cell’s cytosol by diffusion, and certain materials move through the plasma membrane by diffusion (Figure 8.8). Diffusion expends no energy. On the contrary, concentration gradients are a form of potential energy, dissipated as the gradient is eliminated.
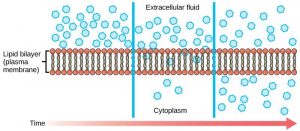
Each separate substance in a medium, such as the extracellular fluid, has its own concentration gradient, independent of the concentration gradients of other materials. In addition, each substance will diffuse according to that gradient. Within a system, there will be different rates of diffusion of the different substances in the medium.
Factors That Affect Diffusion
Molecules move constantly in a random manner, at a rate that depends on their mass, their environment, and the amount of thermal energy they possess, which in turn is a function of temperature. This movement accounts for the diffusion of molecules through whatever medium in which they are localized. A substance will tend to move into any space available to it until it is evenly distributed throughout it. After a substance has diffused completely through a space, removing its concentration gradient, molecules will still move around in the space, but there will be no net movement of the number of molecules from one area to another. This lack of a concentration gradient in which there is no net movement of a substance is known as dynamicequilibrium. While diffusion will go forward in the presence of a concentration gradient of a substance, several factors affect the rate of diffusion.
“Steepness” of the concentration gradient: The greater the difference in concentration, the more rapid the diffusion. The closer the distribution of the material gets to equilibrium, the slower the rate of diffusion becomes.
Mass of the molecules diffusing: Heavier molecules move more slowly; therefore, they diffuse more slowly.
Temperature: Higher temperatures increase the energy and therefore the movement of the molecules, increasing the rate of diffusion.
Solvent density: As the density of a solvent increases, the rate of diffusion decreases. The molecules slow down because they have a more difficult time getting through the denser medium. If the medium is less dense, diffusion increases. Because cells primarily use diffusion to move materials within the cytoplasm, any increase in the cytoplasm’s density will inhibit the movement of the materials. An example of this is a person experiencing dehydration. As the body’s cells lose water, the rate of diffusion decreases in the cytoplasm, and the cells’ functions deteriorate. Neurons tend to be very sensitive to this effect. Dehydration frequently leads to unconsciousness and possibly coma because of the decrease in diffusion rate within the cells.
8.2.3 Facilitated diffusion
In facilitated diffusion, materials diffuse across the plasma membrane with the help of membrane proteins. A concentration gradient exists that would allow these materials to diffuse into the cell without expending cellular energy. However, these materials are ions or polar molecules that are repelled by the hydrophobic parts of the cell membrane. Facilitated diffusion proteins shield these materials from the repulsive force of the membrane, allowing them to diffuse into the cell. These proteins are called transportproteins and can be channels or carrier proteins.
Channels
Channelproteins are transmembrane proteins that fold in such as way as to form a channel or pore through the membrane. Each channel is specific for one particular substance. Channel proteins have hydrophilic domains exposed to the intracellular and extracellular fluids. In addition, they have a hydrophilic channel through their core that provides a hydrated opening through the membrane layers (Figure8.9). Passage through the channel allows polar compounds to avoid the nonpolar central layer of the plasma membrane that would otherwise slow or prevent their entry into the cell. Aquaporins are channel proteins that allow water to pass through the membrane at a very high rate.
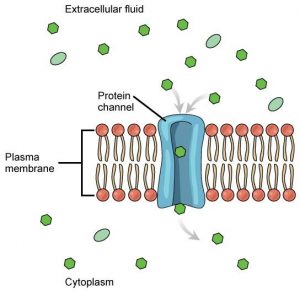
Some channel proteins are always open but many are “gated,” meaning that they can be opened and closed. If a channel is ligand-gated, the attachment of a particular molecule to the channel protein may cause it to open. Other channels are voltage-gated, requiring a change in voltage across the membrane to open them. Cells involved in the transmission of electrical impulses, such as nerve and muscle cells, have voltage-gated ion channels in their membranes.
Carrier Proteins
Another type of transmembrane transporter protein is a carrierprotein. Like channels, carrier proteins are usually specific for particular molecules. A carrier proteins binds a substance and, in doing so, triggers a change of its own shape, moving the bound molecule across the membrane (Figure8.10). Carrier proteins are used to transport molecules that are too large to pass through channels, such as amino acids and glucose.
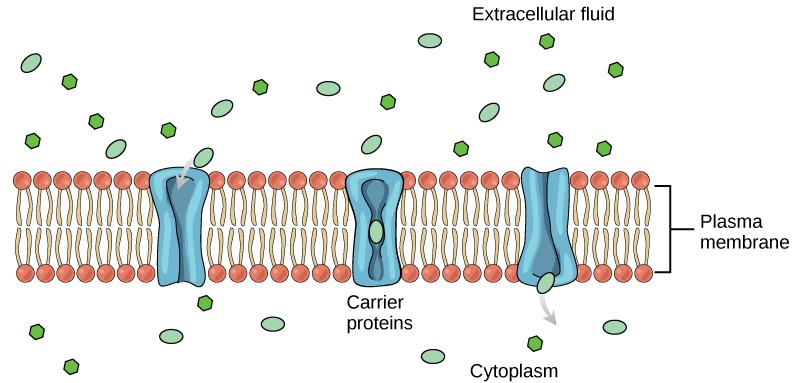
There are a finite number of each type of carrier proteins in any membrane. This can cause problems in transporting enough of the material for the cell to function properly. When all of the proteins are bound to their ligands, they are saturated and the rate of transport is at its maximum. Increasing the concentration gradient at this point will not result in an increased rate of transport.
An example of this process occurs in the kidney. Glucose, water, salts, ions, and amino acids needed by the body are filtered out of the blood in one part of the kidney. This filtrate, which includes glucose, is then reabsorbed in another part of the kidney. Because there are only a finite number of carrier proteins for glucose, if more glucose is present than the proteins can handle, the excess is not transported and it is excreted from the body in the urine. In a diabetic individual, this is described as “spilling glucose into the urine.”
A different group of carrier proteins called glucose transport proteins, or GLUTs, are involved in transporting glucose and other hexose sugars into cells within the body. The hormone insulin, increases the number of GLUTs on cells, causing them to take glucose from the blood when its levels are high. It is this process that is compromised in diabetic individuals.
Channel proteins transport much more quickly than do carrier proteins. Channel proteins facilitate diffusion at a rate of tens of millions of molecules/second, whereas carrier proteins work at a rate of a thousand to a million molecules/second.
8.2.4 Osmosis
Osmosis is the diffusion of water across a semipermeable membrane. Since it is diffusion, it depends on the concentration gradient, or the amount of water on each side of the membrane. The amount of water in a solute is inversely proportional to the concentration of solutes. In other words, the higher the concentration of water, the lower the concentration of solutes, and vice versa. Water can move readily across most membranes, due in part to the presence of aquaporins; however, the membrane limits the diffusion of solutes in the water.
Mechanism of Osmosis
Osmosis is a special case of diffusion. Water, like other substances, moves from an area of high concentration to one of low concentration. An obvious question is what makes water move at all? Imagine a beaker with a semipermeable membrane separating the two sides or halves (Figure8.11). On both sides of the membrane the water level is the same, but there are different concentrations of a dissolved substance, or solute, that cannot cross the membrane (otherwise the concentrations on each side would be balanced by the solute crossing the membrane). If the volume of the solution on both sides of the membrane is the same, but the concentrations of solute are different, then there are different amounts of water, the solvent, on either side of the membrane.
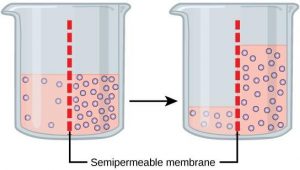
To illustrate this, imagine two full glasses of water. One has a single teaspoon of sugar in it, whereas the second one contains one-quarter cup of sugar. If the total volume of the solutions in both cups is the same, which cup contains more water? Because the large amount of sugar in the second cup takes up much more space than the teaspoon of sugar in the first cup, the first cup has more water in it.
Returning to the beaker example, recall that it has a mixture of solutes on either side of the membrane. A principle of diffusion is that the molecules move around and will spread evenly throughout the medium if they can. However, only the material capable of getting through the membrane will diffuse through it. In this example, the solute cannot diffuse through the membrane, but the water can. Water has a concentration gradient in this system. Thus, water will diffuse down its concentration gradient, crossing the membrane to the side where it is less concentrated. This diffusion of water through the membrane—osmosis—will continue until the concentration gradient of water goes to zero or until the hydrostatic pressure of the water balances the osmotic pressure. Osmosis proceeds constantly in living systems.
8.2.5 Tonicity
Tonicity describes how an extracellular solution can change the volume of a cell by affecting osmosis. A solution’s tonicity often directly correlates with the osmolarity of the solution. Osmolarity describes the total solute concentration of the solution. A solution with low osmolarity has a greater number of water molecules relative to the number of solute particles; a solution with high osmolarity has fewer water molecules with respect to solute particles. In a situation in which solutions of two different osmolarities are separated by a membrane permeable to water, though not to the solute, water will move from the side of the membrane with lower osmolarity (and more water) to the side with higher osmolarity (and less water). This effect makes sense if you remember that the solute cannot move across the membrane, and thus the only component in the system that can move—the water—moves along its own concentration gradient.
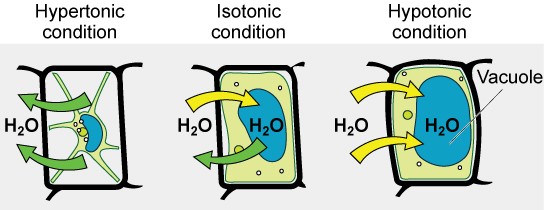
Three terms—hypotonic, isotonic, and hypertonic—are used to relate the osmolarity of a cell to the osmolarity of the extracellular fluid. In living systems, the point of reference is always the cytoplasm, so the prefix hypo- (“lower”) means that the extracellular fluid has a lower concentration of solutes, or a lower osmolarity, than the cell cytoplasm. Blood cells and plant cells in hypertonic, isotonic, and hypotonic solutions take on characteristic appearances (Figure 8.12).
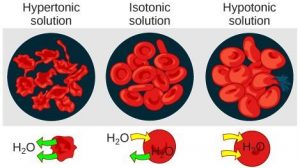
Hypotonic Solutions
In a hypotonic situation, the extracellular fluid has lower osmolarity than the fluid inside the cell. The extracellular fluid has a higher concentration of water than does the cell and water will move down its concentration gradient and enter the cell.
Hypertonic Solutions
In a hypertonic solution (hyper- = “more”), the extracellular fluid has a higher osmolarity than the cell’s cytoplasm. The fluid contains less water than the cell does, so water will leave the cell.
Isotonic Solutions
In an isotonic solution, the extracellular fluid has the same osmolarity as the cell. There is no net movement of water into or out of the cell (although water will still move in and out).
Concept Check
A doctor injects a patient with what the doctor thinks is an isotonic saline solution. The patient dies, and an autopsy reveals that many red blood cells have been destroyed. Do you think the solution the doctor injected was really isotonic?
8.2.6 Tonicity in Living Systems
A red blood cell will burst, or lyse, when it swells beyond the plasma membrane’s capability to expand. In contrast, when excessive amounts of water leave a red blood cell, the cell shrinks, or crenates. Crenation has the effect of concentrating the solutes left in the cell, making the cytosol denser and interfering with diffusion within the cell. The cell’s ability to function will be compromised and it may die. (Figure 8.12).
Living things have ways of controlling the effects of osmosis—a mechanism called osmoregulation. Some organisms, such as plants, fungi, bacteria, and some protists, have cell walls that surround the plasma membrane and prevent cells from lysing. In fact, the cytoplasm in plants is always slightly hypertonic to the cellular environment, and water will always enter a cell if water is available. This inflow of water produces turgor pressure, which stiffens the cell walls of the plant (Figure 8.13). In nonwoody plants, turgor pressure supports the plant. If the plant is not watered, the extracellular fluid will become hypertonic, causing water to leave the cell. In this condition, the cell membrane detaches from the cell wall and constricts the cytoplasm. This process, called plasmolysis, causes plants to lose turgor pressure (Figure 8.14).

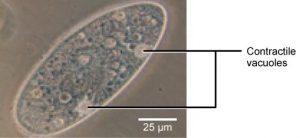
Tonicity is a concern for all living things. For example, paramecia and amoebas, which are protists that lack cell walls, have contractile vacuoles. This vesicle collects excess water from the cell and pumps it out, keeping the cell from lysing as it takes on water from its environment (Figure 8.15).
Many marine invertebrates have internal salt levels matched to their environments, making them isotonic with the water in which they live. Fish, however, must spend approximately five percent of their metabolic energy maintaining osmotic homeostasis. Freshwater fish live in an environment that is hypotonic to their cells. These fish actively take in salt through their gills and excrete diluted urine to rid themselves of excess water. Saltwater fish live in the reverse environment, which is hypertonic to their cells, and they secrete salt through their gills and excrete highly concentrated urine.
In vertebrates, the kidneys regulate the amount of water in the body. Osmoreceptors are specialized cells in the brain that monitor the concentration of solutes in the blood. If the levels of solutes increase beyond a certain range, a hormone is released that retards water loss through the kidney and dilutes the blood to safer levels. Animals also have high concentrations of albumin, which is produced by the liver, in their blood. This protein is too large to pass easily through plasma membranes and is a major factor in controlling the osmotic pressures applied to tissues.
8.3 | Active Transport
By the end of this section, you will be able to:Understand how electrochemical gradients affect ionsDistinguish between primary active transport and secondary active transport
Active transport mechanisms require the use of the cell’s energy, usually in the form of adenosine triphosphate (ATP). If a substance must move into the cell against its concentration gradient—that is, if the concentration of the substance inside the cell is greater than its concentration in the extracellular fluid (and vice versa)—the cell must use energy to move the substance. Some active transport mechanisms move small-molecular weight materials, such as ions, through the membrane. Other mechanisms transport much larger molecules.
8.3.1 Electrochemical Gradient
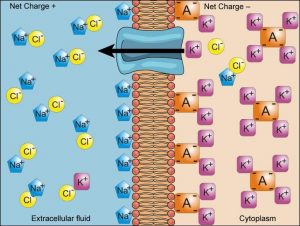
We have discussed simple concentration gradients—different concentrations of a substance across a space or a membrane—but in living systems, gradients are more complex. Because ions move into and out of cells and because cells contain proteins that do not move across the membrane and are mostly negatively charged, there is also an electrical gradient, a difference of charge, across the plasma membrane.
The interior of living cells is electrically negative with respect to the extracellular fluid surrounding them. At the same time, cells have a lower concentration of (Na+) than does the extracellular fluid. Therefore, both the concentration
gradient and the electrical gradient tend to drive Na+ into the cell. Conversely, cells have a higher concentration of K+ than the extracellular fluid does. Therefore, the concentration gradient tends to drive K+ out of the cell, while the electrical gradient tends to drive it inside the cell. The combined gradient of concentration and electrical charge that affects an ion is called its electrochemical gradient (Figure 8.16).
Concept Check
Injection of a potassium solution into a person’s blood is lethal; this is used in capital punishment and euthanasia. Why do you think a potassium solution injection is lethal?
Moving Against a Gradient
To move substances against a concentration or electrochemical gradient, the cell must use energy, usually in the form of ATP. Active transport proteins, called pumps, work against electrochemical gradients. Small substances constantly pass through plasma membranes. Active transport maintains concentrations of ions and other substances needed by living cells in the face of these passive movements. Much of a cell’s supply of metabolic energy may be spent maintaining these processes.
Proteins for Active Transport
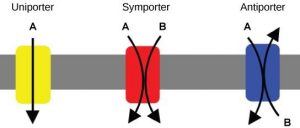
The specific proteins that facilitate active transport are called transporters. There are three types of transporters (Figure 8.17). A uniporter carries one specific ion or molecule. A symporter carries two different ions or molecules, both in the same direction. An antiporter carries two different ions or molecules in different directions. All of these transporters can transport small, uncharged organic molecules such as glucose.
Two mechanisms exist for the transport of small-molecular weight material and small molecules. Primary active transport is directly dependent on ATP. Secondary active transport does not directly require ATP, because it uses electrochemical gradients established by primary active transport for fuel. Primary active transport must occur first to in order to allow secondary active transport to occur. Although it does not use ATP, secondary active transport is still considered active because it requires energy.
8.3.2 Primary Active Transport
One of the most important pumps in animals cells is the sodium-potassium pump (Na+-K+ ATPase), which maintains the electrochemical gradient and the correct concentrations of Na+ and K+ in living cells. The sodium-potassium pump moves two K+ into the cell while moving three Na+ out of the cell (Figure 8.18).
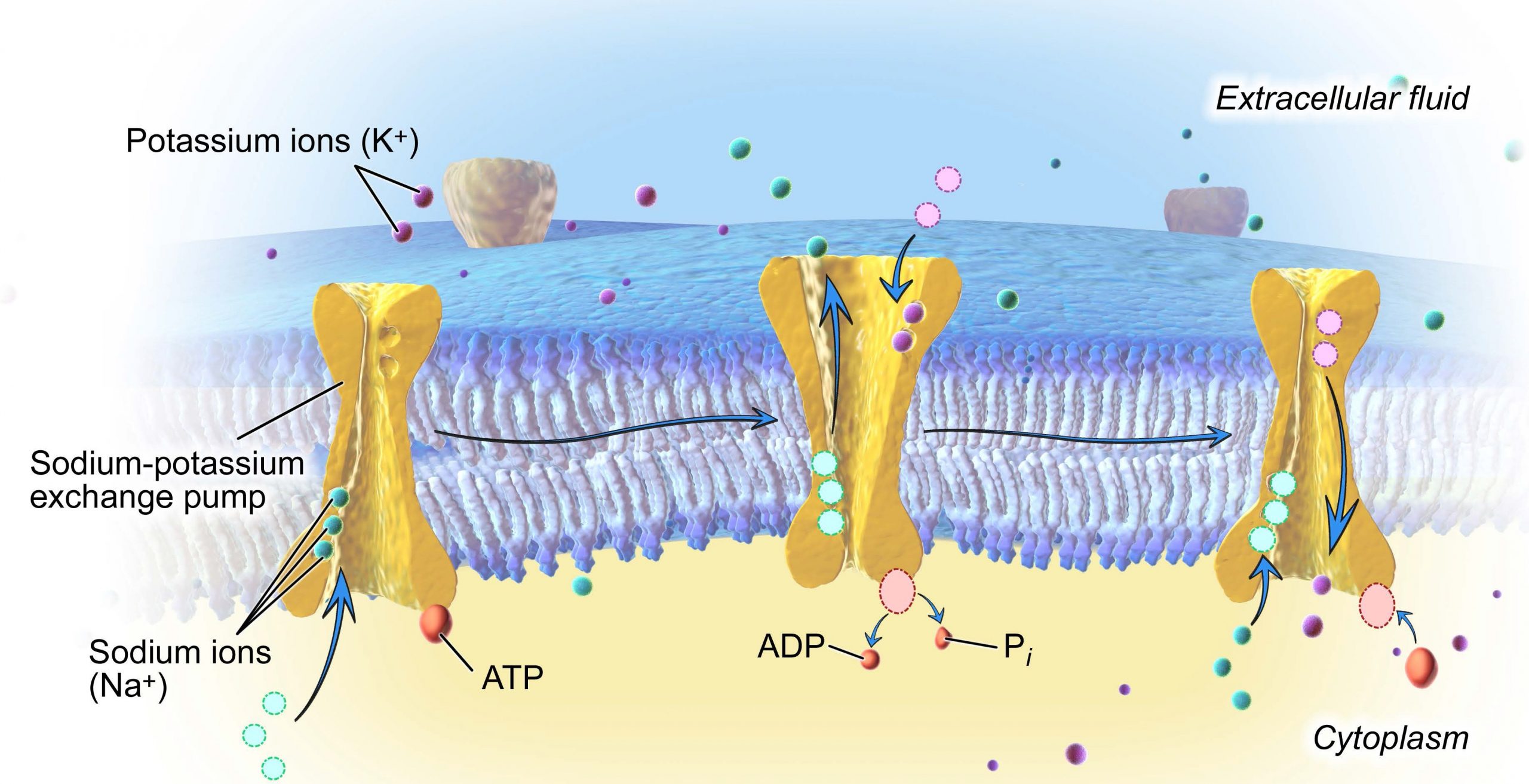
The sodium-potassium pump works in the following six steps:
-
- Three sodium ions bind to the protein.
- ATP is hydrolyzed by the protein carrier and a low-energy phosphate group attaches to it.
- The carrier changes shape and opens towards the exterior of the membrane. The three sodium ions are released.
- Two potassium ions attach to the protein, causing the low-energy phosphate group to detach.
- The carrier protein changes shape so that is open towards the interior of the cell.
- The two potassium ions are released into the cytoplasm and the process begins again.
Several things have happened as a result of this process. First, there are now more sodium ions outside of the cell than inside and more potassium ions inside than out. Second, since three sodium ions moved out for each two potassium ions that moved in, the interior is slightly more negative relative to the exterior. This difference in charge is important in creating the conditions necessary for secondary active transport. The sodium-potassium pump is, therefore, an electrogenic pump (a pump that creates a charge imbalance), creating an electrical imbalance across the membrane and contributing to the membrane potential.
The sodium-potassium pump (Na+/K+ pump) is one example of energy coupling. Each cycle of the Na+/K+ pump moves three sodium out of the cell and brings two potassium into the cell. For each cycle, one ATP is hydrolyzed and its free phosphate group is transferred to the pump protein. This process of a phosphate group binding to a molecule is called phosphorylation. Phosphorylation of the pump protein causes it to change shape, moving ions across the membrane. ATP performs cellular work using this basic form of energy coupling through phosphorylation. Here, the exergonic (energy-releasing) process of ATP breakdown “pays for” the endergonic (energy-requiring) process of moving ions against their concentration gradients.
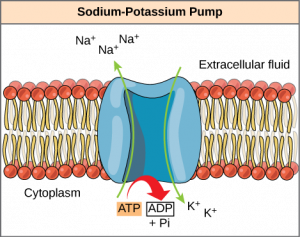
8.3.3 Secondary Active Transport (Co-transport)
Secondary active transport moves a solute against its concentration gradient, an endergonic process, by moving another solute down its concentration gradient, an exergonic process. For instance, as sodium ion concentrations build outside of the plasma membrane because of the action of the sodium-potassium pump, an electrochemical gradient is created. If a channel protein exists and is open, the sodium ions will be pulled through the membrane, down their concentration gradient. This exergonic movement is used to transport other substances that can attach themselves to the transport protein through the membrane (Figure 8.20). Many amino acids, as well as glucose, enter a cell this way.
8.4 | Bulk Transport
Learning Objectives
By the end of this section, you will be able to:
- Describe endocytosis, including phagocytosis, pinocytosis, and receptor-mediated endocytosis.
- Understand the process of exocytosis.
In addition to moving small ions and molecules through the membrane, cells also need to remove and take in larger molecules and particles (see Table 8.2 for examples). Some cells are even capable of engulfing entire unicellular microorganisms. You might have correctly hypothesized that the uptake and release of large particles by the cell requires energy. A large particle, however, cannot pass through the membrane, even with energy supplied by the cell.
8.4.1 Endocytosis
Endocytosis is a type of active transport that moves particles, such as large molecules, parts of cells, and even whole cells, into a cell. There are different variations of endocytosis, but all share a common characteristic: The plasma membrane of the cell invaginates, forming a pocket around the target particle. The pocket pinches off, resulting in the particle being contained in a newly created intracellular vesicle formed from the plasma membrane. The three types of endocytosis are phagocytosis, pinocytosis, and receptor-mediated endocytosis.
Phagocytosis
Phagocytosis (“cell eating”) is the process by which large particles, such as other cells or relatively large particles, are taken in by a cell. For example, when microorganisms invade the human body, a type of white blood cell called a neutrophil will “eat” the invaders through phagocytosis, surrounding and engulfing the microorganism, which is then destroyed by lysosomes inside the neutrophil (Figure 8.21).
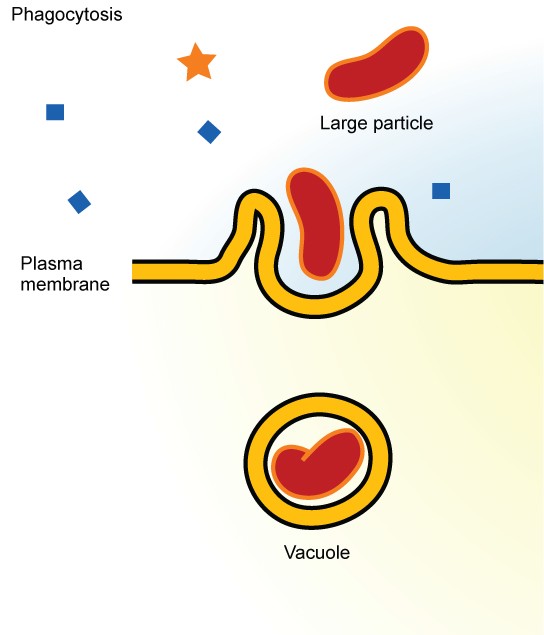
In preparation for phagocytosis, a portion of the inward-facing surface of the plasma membrane becomes coated with a protein called clathrin, which stabilizes this section of the membrane. The coated portion of the membrane then extends from the body of the cell and surrounds the particle, eventually enclosing it. Once the vesicle containing the particle is enclosed within the cell, the clathrin disengages from the membrane and the vesicle merges with a lysosome for the breakdown of the material in the newly formed compartment. When accessible nutrients from the degradation of the vesicular contents have been extracted, the newly formed endosome merges with the plasma membrane and releases its contents into the extracellular fluid. The endosomal membrane again becomes part of the plasma membrane.
Pinocytosis
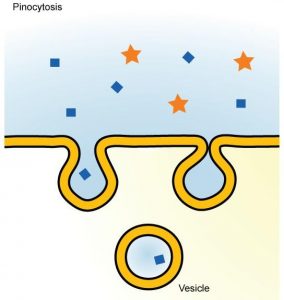
Through pinocytosis (“cell drinking”), cells take in molecules, including water, which the cell needs from the extracellular fluid. Pinocytosis results in a much smaller vesicle than does phagocytosis, and the vesicle does not need to merge with a lysosome (Figure 8.22).
Receptor-mediated Endocytosis
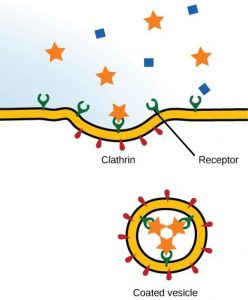
Receptor-mediated endocytosis is a targeted variation of endocytosis that employs receptor proteins in the plasma membrane that have a specific binding affinity for certain substances (Figure 8.23).
Receptor-mediated endocytosis, as in phagocytosis, uses clathrin protein attached to the cytoplasmic side of the plasma membrane. Some human diseases are caused by the failure of receptor-mediated endocytosis. For example, the form of cholesterol termed low-density lipoprotein or LDL (also referred to as “bad” cholesterol) is removed from the blood by receptor-mediated endocytosis. In the human genetic disease familial hypercholesterolemia, the LDL receptors are defective or missing entirely. People with this condition have life-threatening levels of cholesterol in their blood, because their cells cannot clear LDL particles from their blood.
Although receptor-mediated endocytosis is designed to bring specific substances that are normally found in the extracellular fluid into the cell, other substances may gain entry into the cell at the same site. Flu viruses, diphtheria, and cholera toxin all have sites that cross-react with normal receptor-binding sites and gain entry into cells.
8.4.2 Exocytosis
The reverse process of moving material into a cell is the process of exocytosis. The purpose of exocytosis is to expel material from the cell into the extracellular fluid. Waste material is enveloped in vesicle, which fuses with the interior of the plasma membrane, expelling the waste material into the extracellular space (Figure 8.24). Cells also use exocytosis to secrete proteins such as hormones, neurotransmitters, or parts of the extracellular matrix.
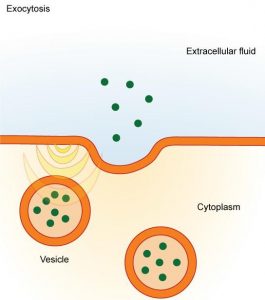
Table 8.2 Methods of transport, energy requirements, and types of material transported
|
Transport Method |
Active/ Passive |
Material Transported |
|
Diffusion |
Passive |
Small-molecular weight material |
|
Osmosis |
Passive |
Water |
|
Facilitated transport/diffusion |
Passive |
Sodium, potassium, calcium, glucose |
|
Primary active transport |
Active |
Sodium, potassium, calcium |
|
Secondary active transport |
Active |
Amino acids, lactose |
|
Phagocytosis |
Active |
Large macromolecules, whole cells, or cellular structures |
|
Pinocytosis and potocytosis |
Active |
Small molecules (liquids/water) |
|
Receptor-mediated endocytosis |
Active |
Large quantities of macromolecules |

